research Interest
Welcome to the Wohl lab!
Our major goal is to fight visual impairment and blindness at the cellular and molecular level. Many retinal diseases, such as age-related macular degeneration (AMD), diabetic retinopathy, glaucoma, retinitis pigmentosa, or myopia, lead to impairment of vision and, in most cases, blindness. Although the impairment of vision is often due to primary damage to the retinal neurons, glial cells play significant roles in this process and can induce secondary neuronal loss. There is increasing evidence that glial cells might also be the cause of retinal disease.
Our lab investigates retinal glia in the healthy and diseased retina and wants to understand their role during disease progression. Our focus lies on Müller glia, the major glial cell type in the retina. We are interested in two major questions:
1) How can we regenerate a diseased retina to restore vision?
2) How can we attenuate disease progression and prevent additional damage to the retina?
Müller Glia - allrounders in the retina
First, what are Müller glia, and why are they so important?
Müller glia are special support cells in the retina, the light-sensitive tissue at the back of the eye that allows us to see. They are the most common support cells in the retina and are created at the very end of retinal development, when all the different neurons of the retina are formed. They were first described in 1851 by Professor Dr. Heinrich Müller, after whom they are named.
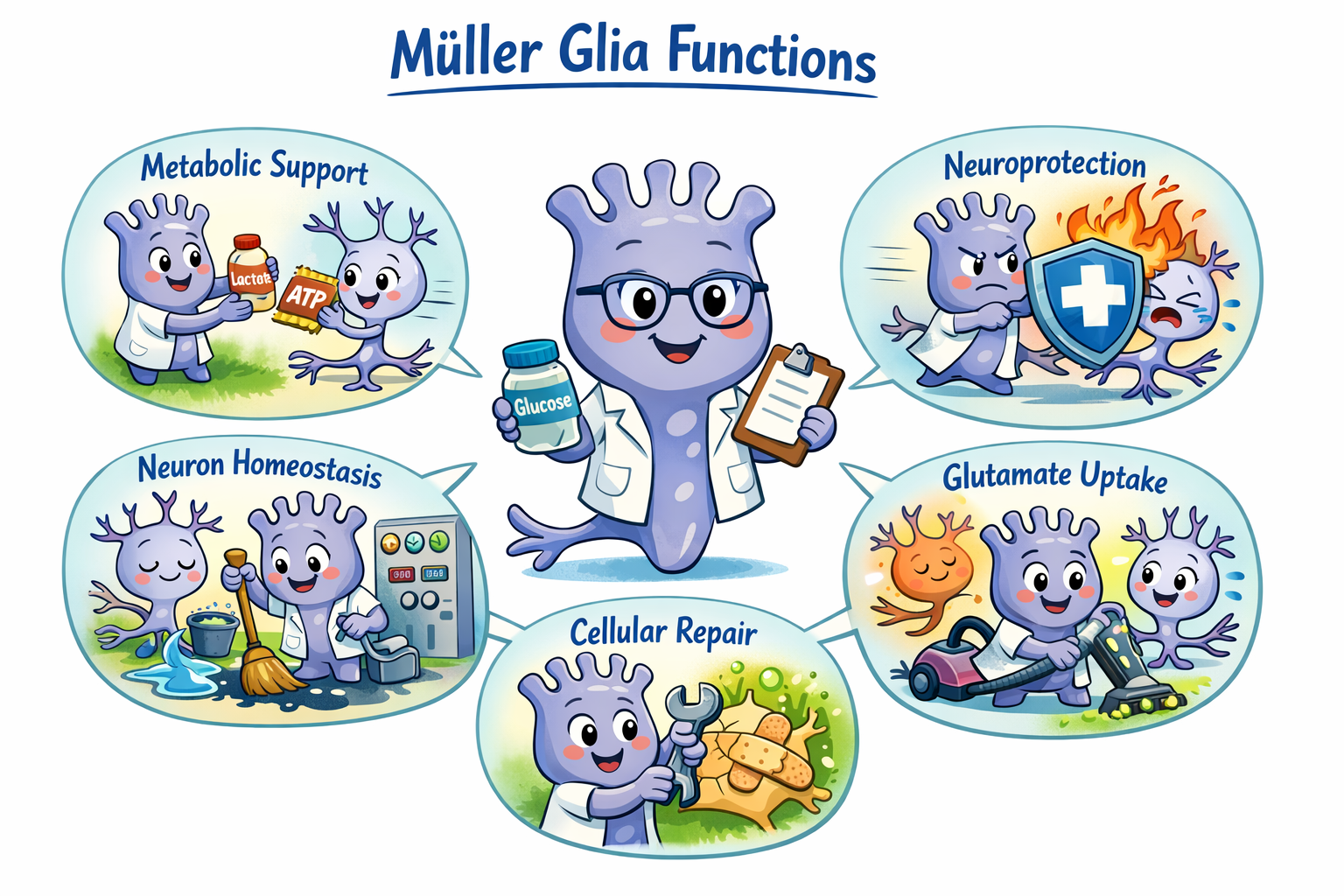
You can think of Müller glia as the “caretakers” of the retina. Just like support cells in the brain, they help keep the retinal environment stable and healthy. They provide nutrients, regulate chemical balance (homeostasis), remove waste, and protect neurons from stress or injury.
Müller glia also help form part of the blood-retinal barrier. This barrier acts like a security system, carefully controlling what can pass from the bloodstream into the retina. Because of this important role, problems with Müller glia are closely linked to diseases that affect retinal blood vessels, such as wet age-related macular degeneration (AMD) and diabetic retinopathy.
In addition, Müller glia are essential for the health and proper function of cone photoreceptors. Cones are the specialized light-sensing cells that allow us to see color and fine detail. They are highly concentrated in the fovea, the small central region of the retina responsible for sharp, detailed vision such as reading and recognizing faces. Because cones depend on Müller glia for metabolic support and stability, the health of these support cells is directly linked to the quality of our central vision.
Our recent research has shown that when Müller glia do not function properly, cone cells are the first to show signs of impairment. This finding suggests that malfunctioning Müller glia may contribute to the early stages, or even the onset, of certain retinal diseases whose origins were previously unclear.
OpenAI-generated cartoon of Muller glia functions.
Emgee (from MG, an abbreviation for Müller glia) is the lab’s mascot.
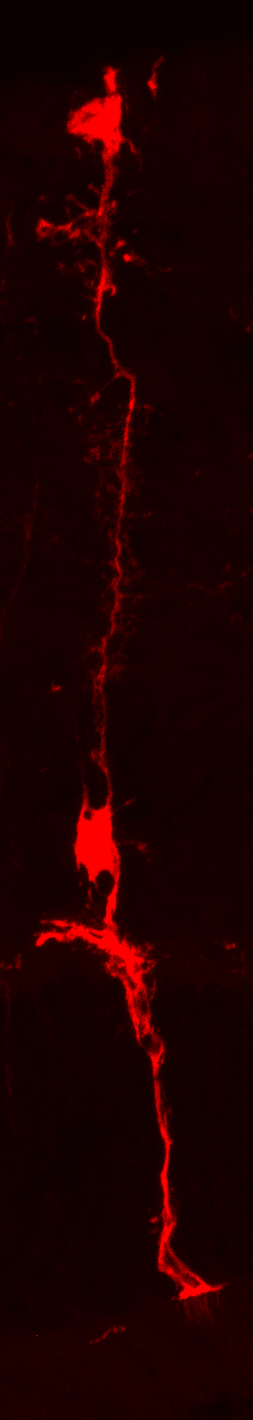
fluorescent mouse Müller glia cell
So, what happens during injury and disease with these cells?
When the retina is damaged or diseased, Müller glia quickly react. They change their shape and their internal activity to form a kind of protective barrier around the injured area. This response, called the reactive state or gliosis, helps contain damage and protect nearby healthy tissue. However, this protective reaction comes at a cost. The barrier they create also blocks regeneration, which is one reason why the human retina cannot naturally repair itself. In some cases, this glial response can even worsen the situation by contributing to inflammation and additional neuronal cell loss (called secondary neuronal loss). It is a complex process that scientists, including ourselves, are still working to fully understand.
Fascinatingly, Müller glia have another remarkable ability, at least in certain animals such as fish. Fish Müller glia can actually repair a damaged retina. When injury occurs, fish Müller glia transform into stem-cell-like cells called progenitor cells. These cells can then divide and generate new, functional neurons to replace the ones that were lost. This allows fish to restore vision after damage. Unfortunately, this regenerative ability is largely switched off in mammals, including humans. However, human Müller glia still retain many molecular features of immature progenitor cells. Researchers believe that, under the right conditions, it may be possible to “reawaken” this dormant potential. One strategy under investigation is called reprogramming. Cellular reprogramming means artificially activating specific factors that are naturally turned on in regenerating fish retinas.
Interestingly, tiny regulatory molecules called microRNAs play important roles in both retinal regeneration in fish and the injury response (gliosis) in mammals. Understanding how these molecules influence Müller glia could open the door to future therapies aimed at repairing the human retina.
microRNAs - tiny powerful regulators with therapeutic potential
MicroRNAs, often called miRNAs, are very small molecules found in every cell of the body. Although tiny, they play a powerful role in keeping cells healthy, including cells in the retina. miRNAs help control how cells grow, develop, and function, including important support cells in the retina called Müller glia.
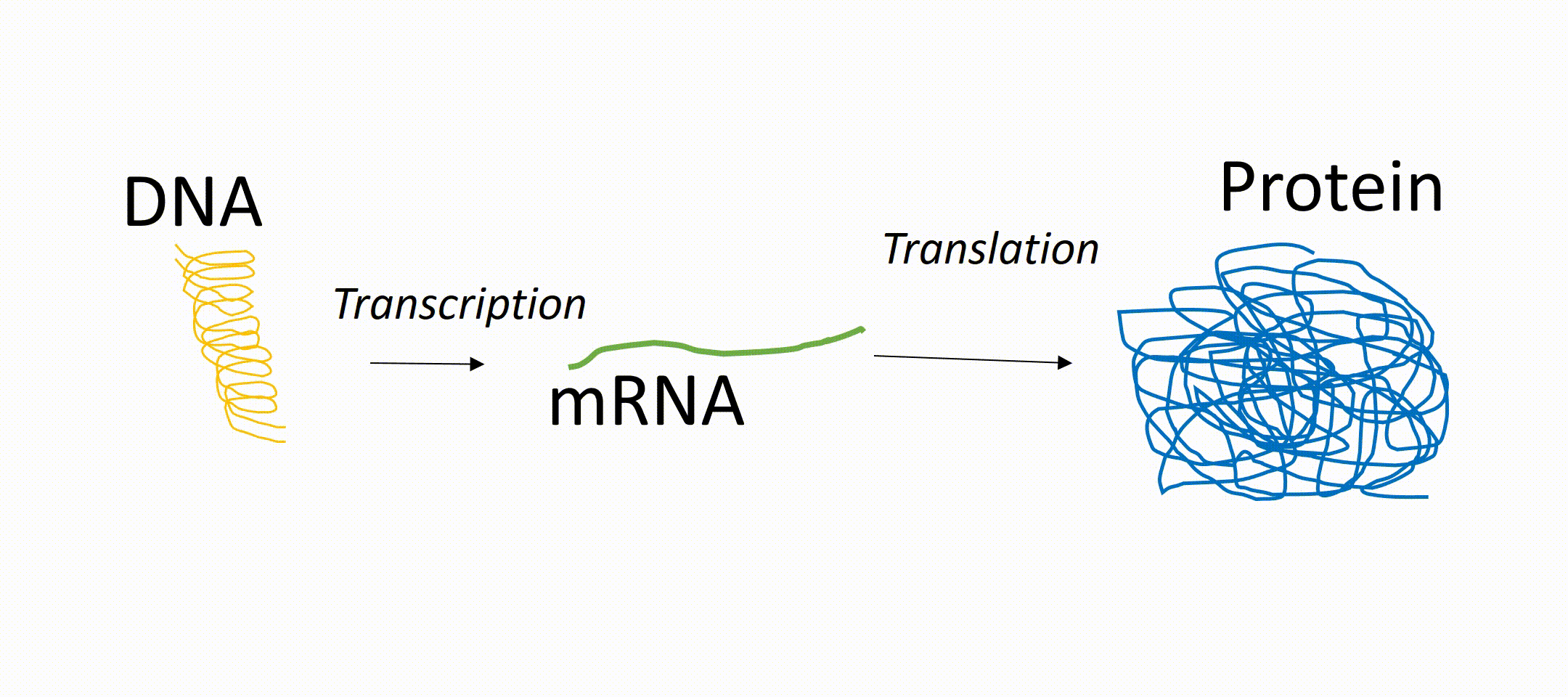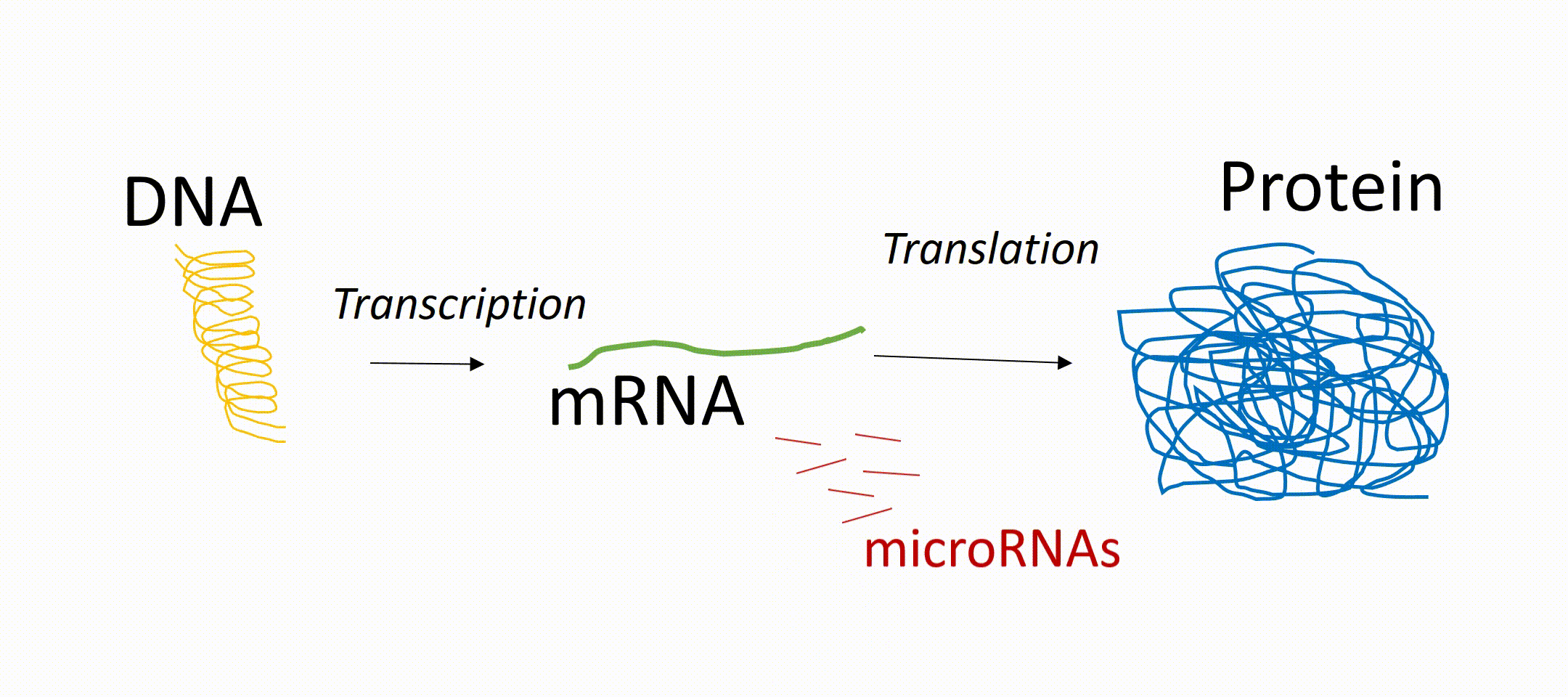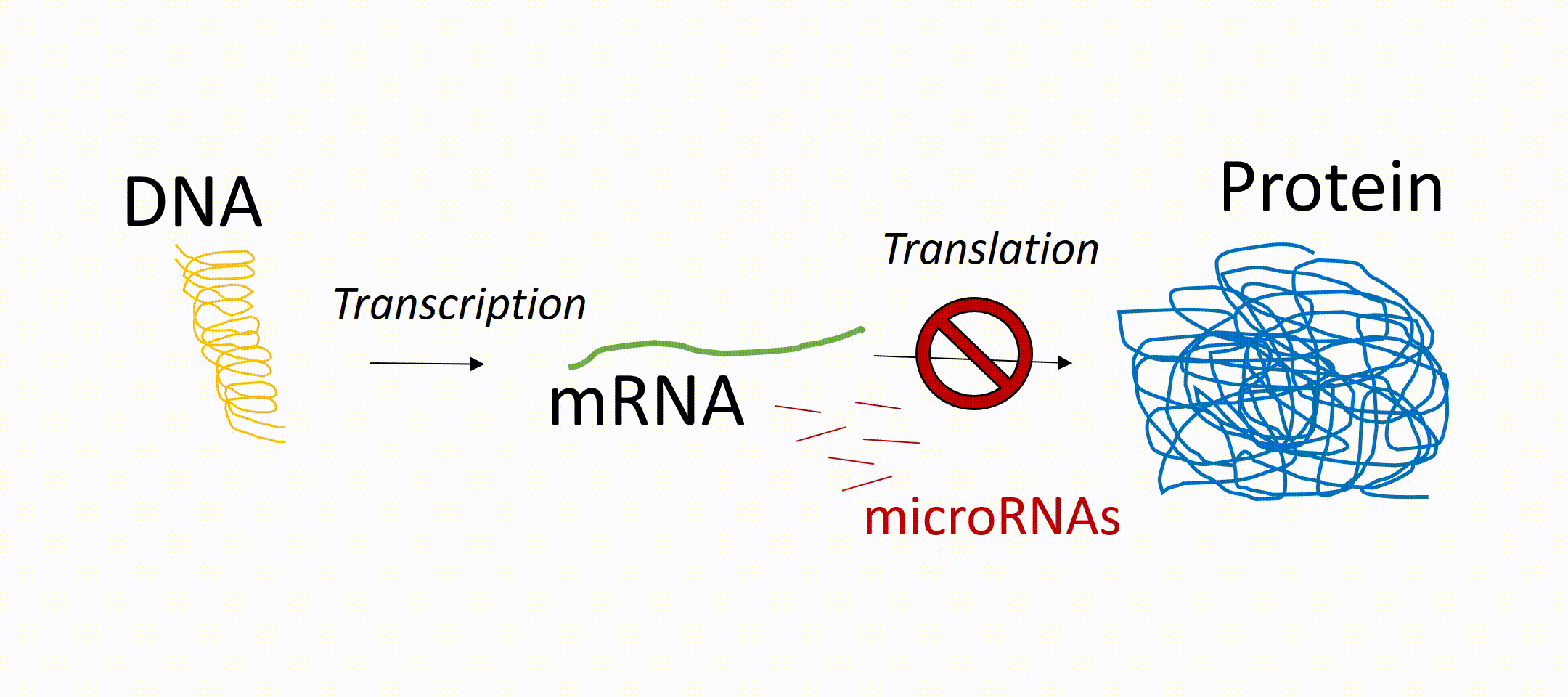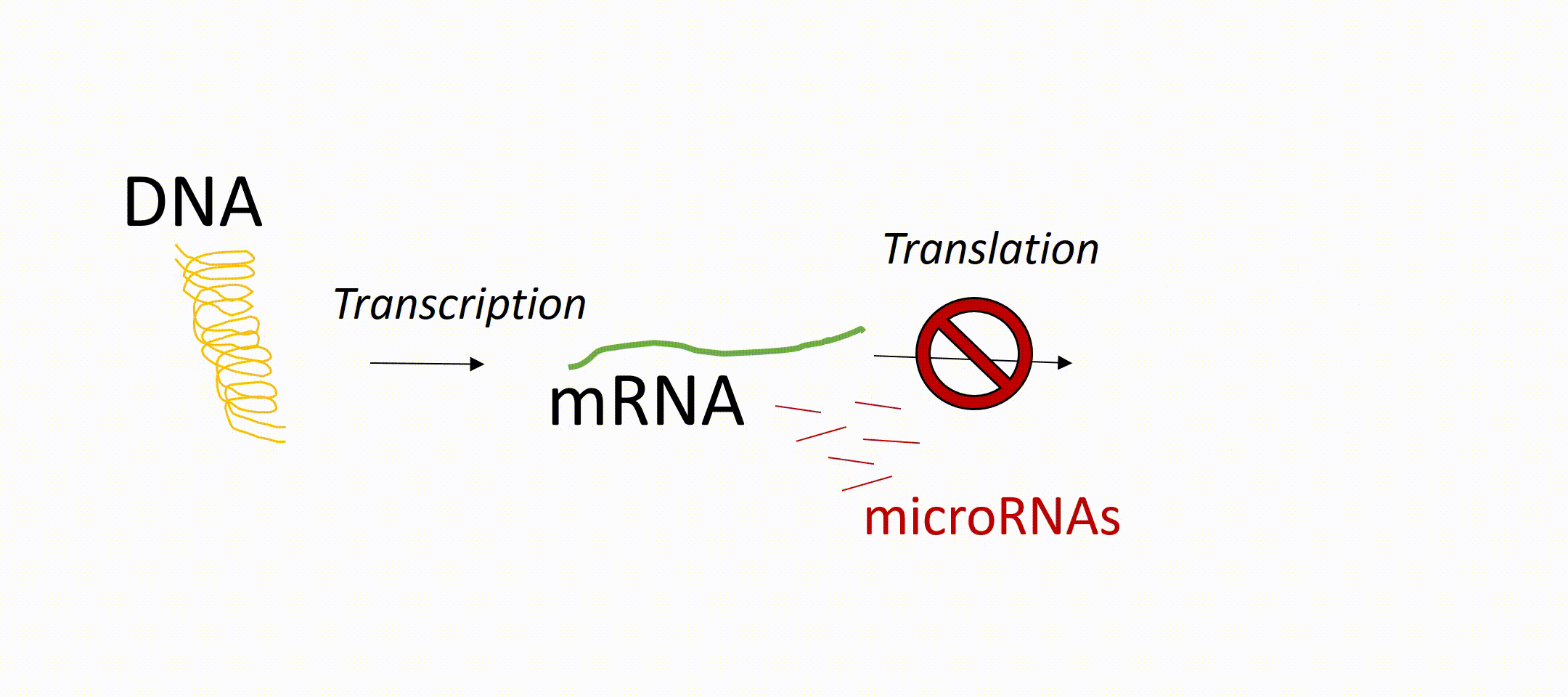
To understand miRNAs, it helps to know how genes work. Genes in our DNA contain instructions for making proteins, which carry out most functions in the body. First, a gene is copied into a message called messenger RNA (mRNA, a DNA transcript). That message is then used to produce a protein. miRNAs act like “dimmer switches.” They can block or reduce the production of proteins, even if the gene itself is active. In this way, they fine-tune how much of a protein is made.
This is important because not all diseases are caused by defective genes. Sometimes the problem is that too much or too little of a normal protein is produced. Since miRNAs control protein production, they offer new possibilities for treatment beyond traditional gene therapy.
There are approximately 1,000 different miRNAs identified so far, and many are nearly identical in mice and humans. This similarity makes them promising tools for developing treatments that can transition from laboratory research to human medicine. However, miRNAs do not behave the same way in every cell or at every stage of life. Their levels can change during development, aging, health, and disease. Therefore, it is crucial to understand which miRNAs are active under specific conditions. This is what we focus on: identifying the sets of miRNAs in the healthy and disease-related retina and during retinal development.
Fascinatingly, as mentioned before, studies in fish have shown that miRNAs help control retinal regeneration. However, this currently occurs only in fish, which, unlike humans, can repair their retinas after injury and restore vision naturally. They do this by inducing a natural program in which Müller glia turn into younger, more immature cells called retinal progenitor cells. These progenitor cells then differentiate into new neurons, which are fully functional. This Müller glial-driven regeneration in fish is regulated by miRNAs. Mammalian Müller glia do not undergo this program after damage, but they can be reprogrammed by means of miRNAs. We showed that miRNA-reprogrammed mammalian Müller glia develop into progenitor cells that differentiate into neurons. We recently discovered that we can produce genuine and functional neurons from reprogrammed young Müller glia under experimental conditions. This insight will help to develop new treatments to stimulate retinal repair in humans in the future, using an endogenous cell source that won’t require transplantation.
But that’s not the only reason why miRNAs are such fascinating molecules. Researchers have also discovered that these small regulators can serve as biomarkers. Biomarkers are measurable indicators of disease, and some of them, including miRNAs, can be detected in blood, saliva, and probably even tears. This could help identify molecular changes even before the retinal cells and the retina itself show pathological signs, enabling early intervention.
microRNA regulation
microRNA as regulators of various events
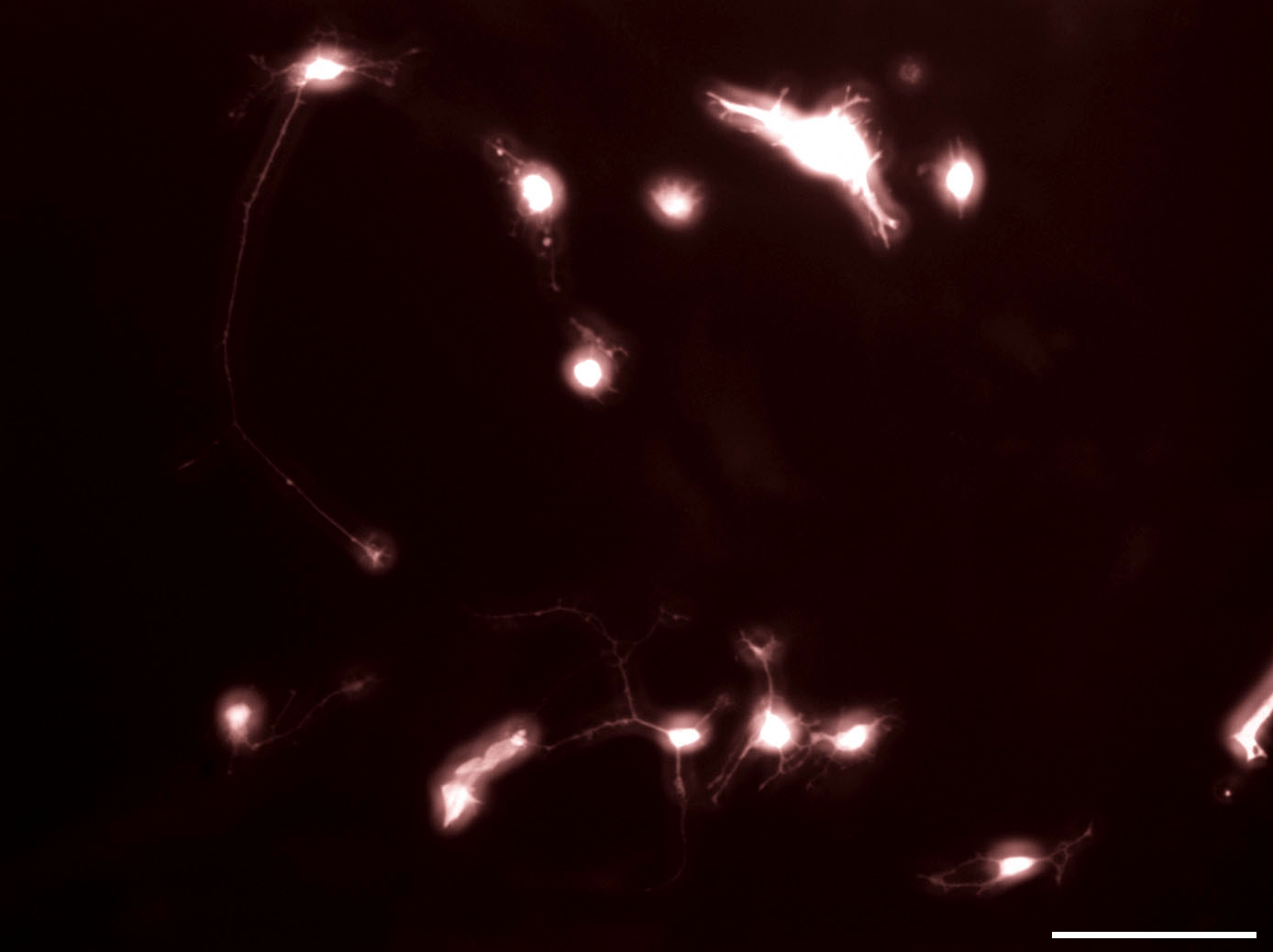
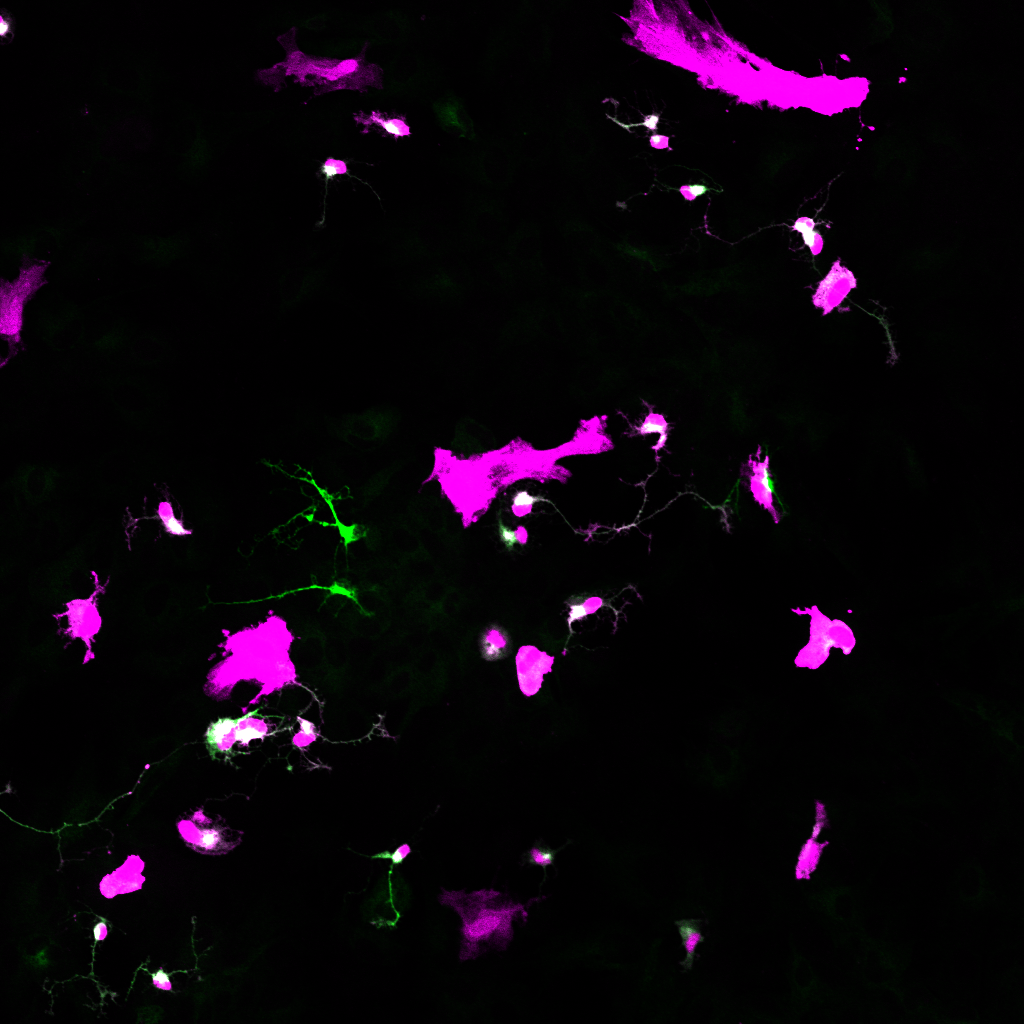
Reprogrammed primary mouse Muller glia after miRNA treatment
The Role of microRNAs in Retinal Glia Function
In my laboratory, we investigate
1) The role of miRNAs in Retinal Development and Müller glia reprogramming. This includes the role of miRNAs during late retinal development in order to understand if miRNAs are required for the generation of late-born retinal cells, which include rod photoreceptors and bipolar cells (interneurons), as well as Müller glia. miRNAs that are required for cell fate development are very likely successful reprogramming factors.
2) The impact of miRNAs in the different phases of glial activation after injury and/or disease.
This will give us a better understanding of the underlying mechanisms of regeneration and gliosis.
The long-term goals are:
1) to utilize microRNAs as a tool for regenerative medicine by stimulating endogenous glia to replace lost neurons.
2) to develop new approaches and therapies to attenuate the glial response after damage to reduce secondary neuronal loss (neuroprotection) and allow potential axonal regeneration of remaining neurons.
Methods and Techniques
Established methods in the lab include:
transgenic mouse models, including retinitis pigmentoa models and in vivo manipulations of microRNAs
primary cell cultures (see protocol published with Jove: https://dx.doi.org/10.3791/63651-v)
immortalized cell line cultures (in vitro)
organotypic explant cultures (ex vivo)
microRNA profiling and analysis: small RNA seq and analysis (bioinformatics)
bulk and single-cell RNA-sequencing including bioinformatics (R, Python)
light damage as a model for dry AMD
optical coherence tomography (OCT) for in vivo imaging
electroretinography (ERG)
protein assays, including ELISA and Western Blot
miRNA:mRNA prediction tools and validation assays, including computation tools such as StarMir, Diana Tools, MirWalk, and dual Luciferase assays
fluorescence microscopy of living cells, including time-lapse, and confocal laser scanning microscopy
and much more.
new insights
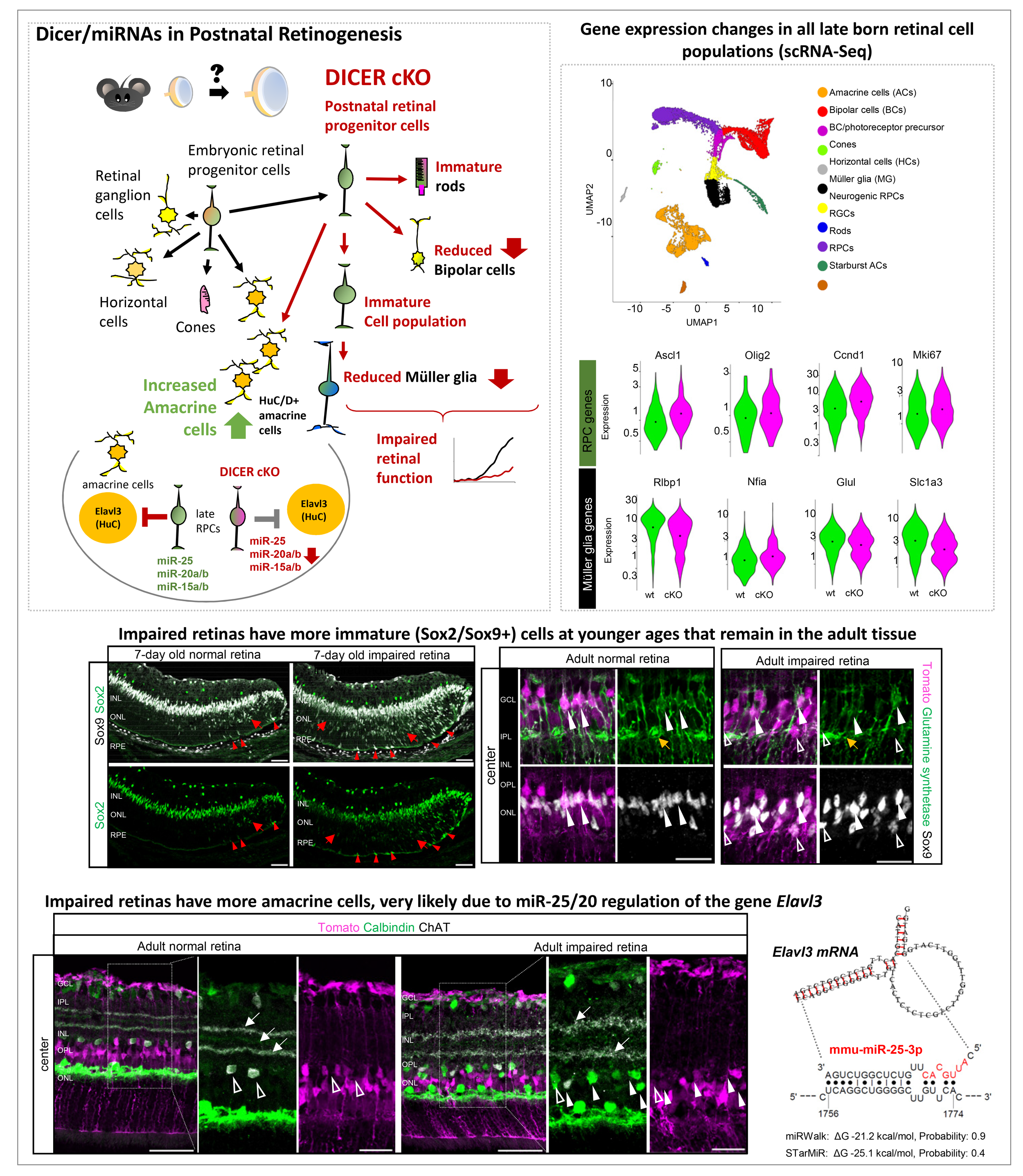
Little regulatory molecules called micro-RNAs are essential for proper maturation, composition, and function in the postnatal retina.
In our recent study published in iScience, we show that altering the molecular composition in immature cells during retinal development results in delays of retinal cell maturation in the developing eye after birth. Specifically, we removed a molecule called Dicer, which produces many small genetic regulators known as microRNAs. These regulators help cells to develop into the right cell types at the right time. When Dicer was removed from these developing retinal cells, the retina did not form properly. In adult eyes, the light-sensing rod cells did not function well, and there were fewer support cells (Müller glia). Interestingly, there was an overproduction of another neuron type called amacrine cells. They are normally born a little earlier, and it appears that their development is regulated by the miRNAs miR-25/20. We also found that some young retinal cells never fully matured and stayed “immature”, i.e., remained stuck in an early developmental stage, even into adulthood. This shows that Dicer and microRNAs are essential for proper retinal cell maturation, so a healthy, functioning retina can form. Our data also shows that miRNAs play a role in Müller glia maturation, hence they can probably be used to rejuvenate these cells for therapeutic purposes.
Why is it important to know that miRNAs play a role in retinal development? Well, knowing which specific miRNAs are regulating a specific cell type will help to develop better tools for endogenous cell reprogramming or for organoid research, two current promising strategies in regenerative medicine to restore lost neurons in patients.
Kang, S., Larbi, D., Bruns, E., Hahne, K., Khodadadi-Jamayran, A., Sreenivasaiah, C., Lima Carneiro, M., Andrade, M., Batsuuri, K., Chen, S., Jager, J., Viswanathan, S., Clark, B.S., Wohl, S. G. 2025. “Dicer is essential for proper maturation, composition, and function in the postnatal retina.” iScience, 28, 113794.
Dicer/miRNAs are essential for proper maturation, composition, and function in the postnatal retina. Kang et al., iSciene 2025, https://doi.org/10.1016/j.isci.2025.113794
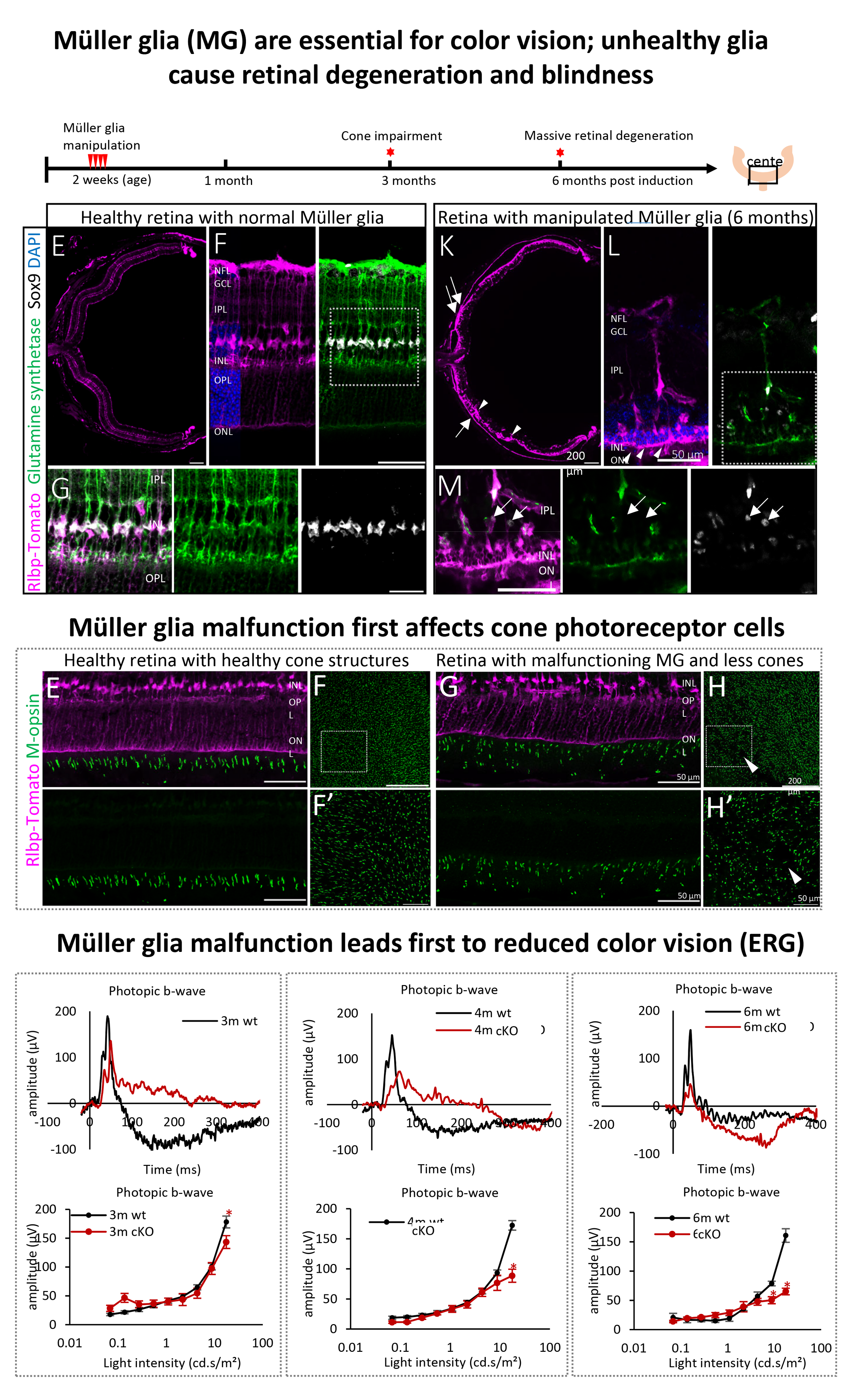
Müller glia ensure proper cone Photoreceptor function and therefore ensure color vision
In another recent study published in IOVS, we show that altering the molecular composition in young Muller glia results in the loss of their physiological, essential properties. The first affected neuron type is cone photoreceptors, which are required for color vision. When we removed a key enzyme called Dicer, which helps produce small gene regulators (microRNAs), the cones began to lose their structure and function. Over time, the damage progressed and resulted in severe retinal degeneration.
This study shows that Müller glia are not just passive support cells, but essential key elements critical for maintaining healthy vision. Importantly, we demonstrate for the first time that malfunctioning Müller glia alone can trigger retinal degeneration that resembles diseases such as retinitis pigmentosa or dry age-related macular degeneration.
Why is that important to know? Well, understanding that specific microRNAs are necessary to keep glial cells functioning properly reveals a new possible cause of retinal disease. Instead of focusing only on damaged neurons, we now see that problems in support cells can start the disease process. This opens the door to new treatment strategies aimed at restoring the normal balance of microRNAs in glial cells. By doing so, we may be able to slow disease progression and delay vision loss.
Larbi, D., Rief, A.M., Kang, S., Chen, S., Batsuuri, K., Fuhrmann, S., Viswanathan, S., Wohl, S. G., 2025. Dicer Loss in Muller Glia Leads to a Defined Sequence of Pathological Events Beginning With Cone Dysfunction. Invest Ophthalmol Vis Sci, 66, 7.
Histology and electroretinography of normal retinas and retinas with manipulated Muller glia.
PREVIOUS work
microRNA sets in Müller glia, neurons, and retinal progenitor cells (RPCs).
1: Müller Glia have a specific set of mircoRNAs
Müller glia have a specific set of microRNAs different from neurons and progenitor cells. However, besides distinct miRNA sets, there are also miRNAs that Müller glia share with other cell types.
Wohl, S. G. and Reh, T.A., 2016. The microRNAs expression profile of mouse Müller glia in vivo and in vitro. Scientific Reports 6, 35423; doi 10.1038/srep35423.
Wohl et al., 2019. MicroRNAs miR-25, let-7 and miR-124 regulate the neurogenic potential of Muller glia in mice. Development 146;10.1242/dev.179556
2: microRNAs can convert Müller glia into retinal Progenitors
The microRNAs neuronal microRNA miR-124 (in combination with miR-9 and miR-9*) can reprogram Müller glia into Ascl1+ retinal progenitor cells that give rise to retinal neurons. The combination of miR-124/9/9* together with the transcription factor Ascl1 accelerates reprogramming. Moreover, over-expression of the retinal progenitor microRNA miR-25 and inhibition of the Müller glia microRNA let-7 leads to conversion of Müller glia into retinal progenitors and subsequent neuronal differentiation.
Wohl, S. G. and Reh, T.A., 2016. miR-124-9-9* potentiates Ascl1-induced reprogramming of cultured Müller glia. Glia 64, 743-762.
Wohl, S. G., Hooper M. and Reh, T.A., 2020. MiroRNAs miR-125, let-7 and mIR-124 regulate the neurogenic potential of Müller glia in mice. Development, 146.
microRNAs convert primary Müller glia into Ascl1 expressing retinal progenitor cells that differentiate into network-forming neurons.
3: microRNAs are essential for Müller glia function and overall retinal health
The depletion of microRNAs in Müller glia (by deleting Dicer, the enzyme that generates mature microRNAs) leads to significant disruptions in the retinal architecture. Müller glia proliferate, migrate and form strange aggregations but do not function properly anymore. As a consequence, photoreceptors die and a phenotype that resembles retinitis pigmentosa can be observed over time. Experiments using organotypic explants cultures show that supplementation with miRNAs helps to rescue the Müller glia phenotype and overall retinal architecture. This implies that dysregulation of miRNAs in Müller glia could be an additional cause of degenerative diseases and consequently, treating diseased retinas with miRNAs could help rescue the tissue.
Wohl, S. G., Jorstad, N. L., Levine, E., and Reh, T.A., 2017: Müller glial microRNAs are required for the maintenance of glial homeostasis and retinal architecture. Nature Communications, 8(1):1603. DOI: 10.1038/s41467-017-01624
4: Reactive Müller glia lose miRNAs
Light damage is a model to induce massive degeneration as it occurs during retinitis pigmentosa or AMD. This injury model leads, similar to other damage paradigms, to reactive gliosis. We found that Müller glia in severely damaged retinas (10,000 lux, 8 h) have significantly reduced levels of miRNAs. This profile resembled the profile we obtained after miRNA depletion (Dicer-cKO mouse). Only a few miRNAs were found to be upregulated after damage. Interestingly, known upregulated generic gliosis genes such as GFAP and Cxcl10 were found to be targeted by Müller glia miRNAs. Moreover, we found an upregulation of stress genes including Maff and Atf3, in both, reactive glia after light damage and mRNA-depleted Müller glia, that were also found to be regulated by Müller glia miRNAs. This suggests that miRNA treatments (supplementation) could attenuate the glial stress response/gliosis that occurs during degenerative retinas.
Kang, S., Larbi, D., Andrade M.D., Reardon, S., Reh, T.A., Wohl, S.G., 2020. A comparative analysis of reactive Müller glia gene expression after light damage and microRNA-depleted Müller glia – focus on microRNAs. Frontiers in Cell and Developmental Biology: 620459 doi: 10.3389/fcell.2020.620459
LAB Members
Email: swohl@sunyopt.edu
phone: +1 212 938 5822
Stefanie G. Wohl, Principal Investigator
I was born in Germany and studied Biology (Diploma) at the Friedrich Schiller University (FSU) of Jena, Germany. Neurobiology, regeneration, and stem cell research were always topics I was interested in and fascinated by. In 2003, I started working as an undergraduate in the laboratory of Dr. Stefan Isenmann, M.D., at the Clinic of Neurology (FSU) in Jena, which investigated axonal regeneration of the optic nerve. I continued my work as a graduate student under his supervision as well as that of Dr. Christian Schmeer, Ph.D., and Dr. Jürgen Bolz, Ph.D. Dr. Bolz was my professor of neurobiology, who was trained by Dr. Heinz Wässle, Ph.D. My research topic was the identification of putative stem cell-like cells after moderate and severe injury. This included studies of neurodegeneration, gliosis, and immunological cell responses. At this time, I became intrigued with Müller glia in the retina via the work of Dr. Andreas Reichenbach and Dr. Andreas Bringmann from the University of Leipzig. Although my focus was on Müller glia, by coincidence, I discovered a subtype of microglia that transiently increased in number after optic nerve injury, expressing a marker that was primarily associated with stem/ progenitor cells (nestin). So my studies took a little detour by characterizing endogenous retinal microglia after optic nerve injury.
In 2011, I received my Ph.D. (neuroscience/ ophthalmology) with the highest honors (summa cum laude) from the Friedrich Schiller University of Jena. For my post-doc training, I decided to go abroad and joined the laboratory of Tom Reh at the University of Washington in Seattle. During this time, I discovered my interest in and passion for microRNAs and focused my research again on Müller glia. We profiled the set of microRNAs in the glia at different ages, in vitro and in vivo, as well as in retinal progenitor cells. We also developed a mouse in which Müller glia lose their microRNAs over time to study the impact of these molecules on glial cell state and function. These studies formed the foundation of my current work.
In September 2018, I became an Assistant Professor at the State University of New York, College of Optometry in the Department of Biological and Vision Sciences and founded my laboratory, the first molecular biology lab in the College. Since 2024, I have been a tenured Associate Professor, and our research focus is on understanding the role of microRNAs in retinal development and Müller glia function, and utilizing these molecules as potential therapeutic tools to fight blindness.
I was a recipient of a Research Fellowship from the German Research Foundation (DFG, 2014-2016) and the SUNY Empire Innovation Grant (2018-2022). Since 2022, my research has been funded by the NIH (R01EY032532).
Monica Andrade, Vision Research Coordinator
Monica joined the lab in 2019 as a Senior Research Support Specialist. She was promoted to Vision Research Coordinator in 2021 but is still affiliated with the lab. She received her Bachelor of Arts from Rutgers University in New Brunswick, NJ with Biology as a major. She received a Master of Science from Long Island University, Brooklyn campus, with Medical Microbiology as a specialty.
She worked in multiple projects involving epigenetic studies of the transcriptional repressor Zinc finger protein 57homolog (ZFP57), characterization and biological function of recombinant tissue factor pathway inhibitor beta (TFPIß), and the construction of 16S rRNA libraries for studies of the human microbiome.
email: mandrade@sunyopt.edu
phone: +1 212 938 5822
Luke Gibbons, PhD, postdoc
Luke received his PhD in 2023 from Trinity College Dublin in Ireland, under the supervision of Prof. Sarah Doyle. His research focused on characterizing the role of the protein SARM1 in the induction of photoreceptor cell death and immune cell recruitment in mouse models of retinal degenerative disease. Additionally, he evaluated a SARM1-targeting antisense oligonucleotide as a potential therapy in mouse models of dry AMD and retinitis pigmentosa.
Following completion of his PhD, Luke remained in the Doyle lab at TCD as a postdoctoral researcher, continuing his work on SARM1 and assisting with an in vivo study examining the therapeutic potential of immune cell transfers for the treatment of wet AMD.
Luke joined the Wohl lab as a postdoctoral associate in October 2025. He is interested in examining the role of miRNAs in retinal development and disease with a focus on models of dry AMD and retinitis pigmentosa.
email: lgibbons@sunyopt.edu
Shaoheng (Raven) Chen OD, Residency/MS Student
Raven received his Doctor of Optometry from SUNY in 2024 and is a licensed optometrist in New York. In 2020, he received a Bachelor of Medicine from Wenzhou Medical University, where he gained experience in developing a real-time imaging platform of human oxygen metabolism with visible spatially modulated light.
Raven was accepted in combined Master and Residency program 2024-2026 at SUNY Optometry. He has a strong motivation to integrate basic science into clinical Optometry. His project is to study the impact of Müller glia microRNAs in a mouse model of retinitis pigmentosa.
email: schen6@sunyopt.edu
Alexander Rief, OD, MS
Alex received his Bachelor of Science (BS) from the University at Albany while being enrolled in the 3+4 Joint Program with the SUNY College of Optometry. After starting the graduate program, he was accepted into the Dual OD/MS Program at the end of 2021. He then joined the Wohl Lab at the start of 2022. Alex has an interest in the field of ocular disease and his research is focused on characterizing Muller cells in a retinal degeneration model replicating retinitis pigmentosa. He has been awarded the T35 Research Grant in the summer of 202 and the Dr. Joshua Wallman Memorial Award for Research in May 2025.
Alex successfully graduated in May 2025 and is still affiliated with the lab during his current residency.
email: arief@sunyopt.edu
Katarina Gotouhidis, BS
Katerina received her Bachelor of Science from The American University in 2025, where she studied biology on the premedical track. Her interests in ocular health and disease began there, where she developed her understanding of molecular and developmental biology.
Throughout her undergraduate years, she supplemented her education with diverse clinical experience, working as a medical assistant at Foxhall Podiatry Associates and Westport Chiropractic and Acupuncture.
She began her studies at SUNY College of Optometry as an OD candidate in 2025, where she quickly took an interest in the retina and ocular disease. She began volunteering as a member of the Wohl Lab in the winter of 2025. Katerina aims to integrate her research interests into her future career as an optometrist, providing comprehensive evidence-based care to her patients.
Madelayne Shadkam, BS
Madelayne is a first-year Doctor of Optometry (OD) student at SUNY College of Optometry and is expected to graduate in 2029. She graduated from California State University, Monterey Bay, in 2024 with a Bachelor of Science in Molecular Biology and a minor in Chemistry.
Originally from Los Angeles, she moved to the East Coast to pursue her long-standing goal of studying optometry in New York. Madelayne joined the lab in February 2026 to begin assisting with research, as she has always been passionate about integrating her interest in biology with her clinical training. She was particularly drawn to the lab’s research on microRNAs and their role in retinal function and disease, with the goal of better understanding how these mechanisms contribute to blindness. She believes that participating in research alongside her clinical education will help her become a more well-rounded optometrist.
Emily Yu, BS
Emily earned her Bachelor’s in Biology from the College of William and Mary in May 2024 and is currently working as veterinay assistant at Brooklyn Heights Veterinary Hospital while preparing for her next chapter in academia.
Emily has research experience studying neurodegeneration of dopaminergic cells in zebrafish during her undergraduate years and previously worked as a translational research assistant focused on pancreatic cancer. She has an interest in Müller glia and retinal disease mechanisms and joined the lab in Winter 2025 as an intern to assist with experiments, data collection, or other lab support.
Alumni
Ashley Indictor, BS, Intern
Daniel Larbi, O.D., Ph.D., PhD student, recipient of the Trainee Professional Development Award (TPDA) from the Society for Neuroscience (SfN), 2023
Seoyoung Kang, MMsc, Ph.D., PhD student, recipient of the Minnie and Roseanna Turner Fund for Impaired Vision Research 2023
Mariana Lima Carneiro, BS, MS, Intern and Research Technician
Khulan Batsuuri, MD, PhD, postdoc
Konstantin Hahne, BS, MS, Master’s student (Optometry), Berliner Hochschule für Technik
Eik Bruns, BS, MS, Master’s student (Optometry), Berliner Hochschule für Technik, DAAD stipend recipient
T-35 and SUNY SUMMER STIPEND RECIPIENTS
Annelise Cassidy, O.D. cand., SUNY-Optometry (2025)
Faiz Khan, O.D. cand., SUNY-Optometry (2024)
Maxine Humphrey, O.D. cand., SUNY-Optometry (2024)
Julia Jager O.D. cand., SUNY-Optometry (2023)
Alexander Rief O.D. cand., SUNY-Optometry (2022)
Quynh Nguyen O.D. cand., UC Berkeley School of Optometry (2019)
Gallery: Starting the lab - Lab members over the years - Conferences over the years - Lab fun events over the years ..


















Join us!
Are you interested in becoming part of our team? We are currently seeking a highly motivated team member with a strong molecular biology/genetics background. Interns/trainees with interest in cellular and molecular vision science are also very welcome to join our lab!
For more info about the PhD student programs see https://www.sunyopt.edu/education/admissions/graduate_programs
For general inquiries, please use the contact form below. For an application, please sent your letter and detailed CV to swohl@sunyopt.edu.
location
The State University of New York
College of Optometry
Department of Biological and Vision Sciences
33W 42nd Street 10036, New York, NY
Email: swohl@sunyopt.edu
Phone office: +1 212 938 4069
Phone lab: +1 212 938 5822
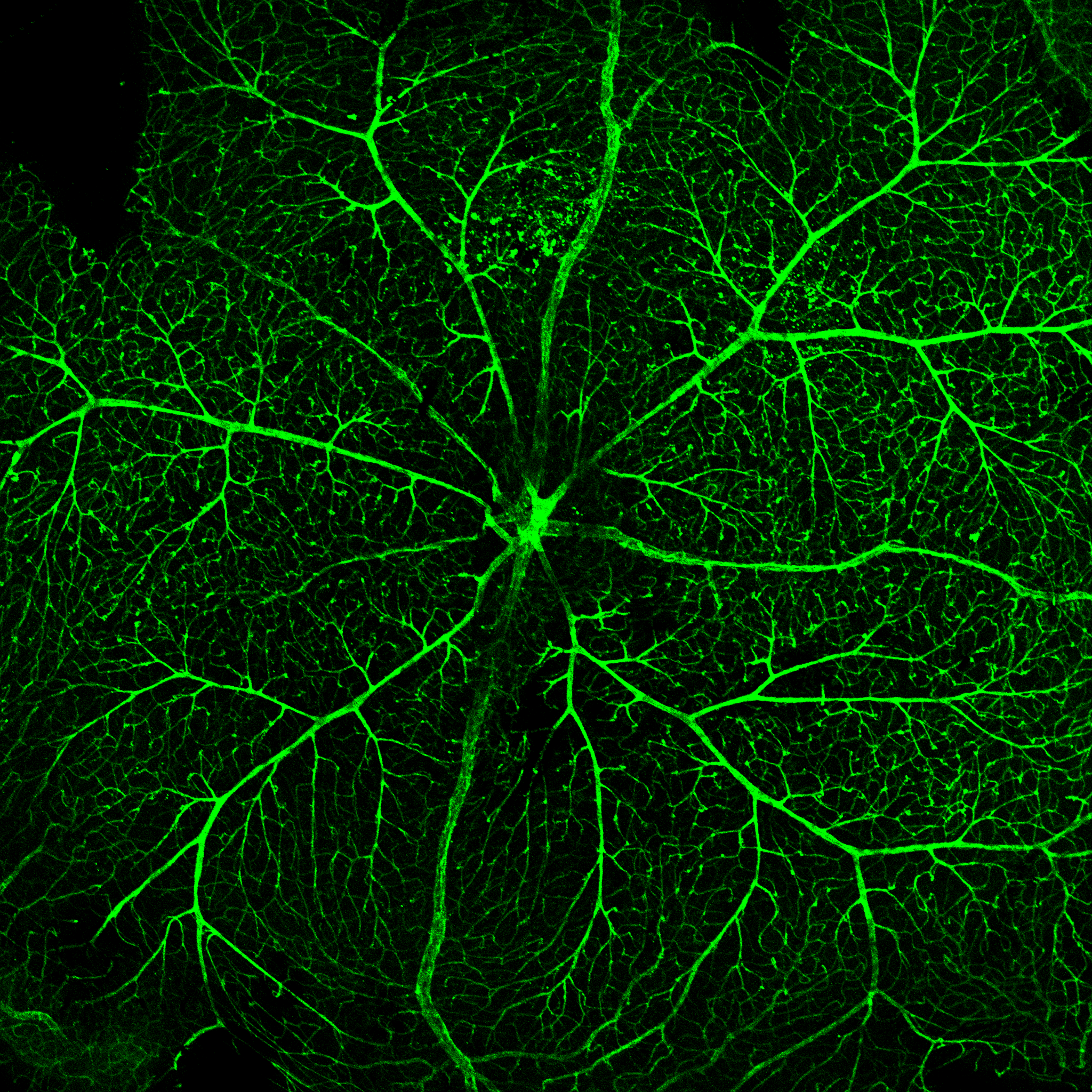
Gallery: the beauty of retinal cells and Cells of the eye - in vivo and in vitro - Visualized via fluorescent colors









Publications
Books
“Neuroscience and Biobehavioral Psychology” Chapter “Müller glia Development” Wohl S.G., Editor in Chief: Patricia d’Amore, Section Editor: Nadean Brown. Section Title: Ocular Development. https://doi.org/10.1016/B978-0-443-13820-1.00126-2
"Molecular Therapies for Inherited Retinal Diseases" Chapter “Retinal miRNA functions in health and disease.” Zuzic M., Rojo Arias J. E., Wohl S.G., and Busskamp V., reprint. https://doi.org/10.3390/books978-3-03943-177-9, ISBN 978-3-03943-176-2 (Hbk); ISBN 978-3-03943-177-9 (PDF), published: October 2020